While chest compressions and medications are frequently studied in cardiac arrest trials, one essential component is often neglected: manual ventilation. Inconsistent and unmeasured, it introduces variability that could impact outcomes. It’s time to shine a light on this overlooked factor—and explore how feedback tools can help.
A call for standardization with Ventilation Feedback Devices
Over the last decade, CPR research has delivered important breakthroughs—from better drugs and improved chest compressions to cutting-edge techniques like extracorporeal resuscitation. Influential studies like PARAMEDIC2, ARREST, and INCEPTION have helped shape international resuscitation guidelines and clinical protocols.
But there is a major piece of the puzzle that has often been ignored: ventilation. Most large-scale trials have failed to monitor or control how ventilation is performed during CPR. This is introducing a hidden layer of variability—what we might call a “ventilation bias.” This oversight can influence outcomes and may even mask the true value of interventions under study.
The ventilation bias in CPR trials
Even in some of the most rigorous CPR studies to date, ventilation hasn not received the attention it deserves:
- PARAMEDIC2, which evaluated the effects of epinephrine in out-of-hospital cardiac arrest, didn’t track tidal volume or ventilation rate. Teams followed standard practice, but without feedback or measurement, ventilation quality likely varied widely—potentially affecting neurological outcomes.
- In the ARREST trial, which focused on the use of extracorporeal CPR (ECPR) for refractory ventricular fibrillation, the quality of ventilation during the conventional CPR phase before ECMO was neither reported nor standardized. That leaves a critical data gap, as inconsistent ventilation could have influenced the results.
- Similarly, INCEPTION, a multicenter trial comparing ECPR to conventional care, offered minimal information on how ventilation was delivered or monitored during CPR. Without this data, it’s difficult to interpret whether differences in outcomes stem from the interventions or from differences in basic life support, including ventilation.
Across these landmark studies, ventilation remains an unmeasured and uncontrolled variable.
Why manuel ventilation matters ?
Effective ventilation during CPR is crucial for two things: delivering oxygen and clearing carbon dioxide. Both are essential for achieving return of spontaneous circulation (ROSC) and for protecting the brain. But hypoventilation—not giving enough air, or going too long between breaths—is surprisingly common, especially in high-stress situations like prehospital cardiac arrests.
This is not just a theoretical issue. A pivotal study led by Ahamed Idris found that patients who received adequate ventilation were three times more likely to survive to hospital discharge, and four times more likely to recover with good neurological function, compared to those who were hypoventilated. These are not small differences—they are clinically significant, with real implications for patient outcomes.
And yet, in most clinical practice and research, ventilation is still done by feel, without any real-time feedback to guide the provider. That leaves plenty of room for underperformance—often without anyone realizing it.
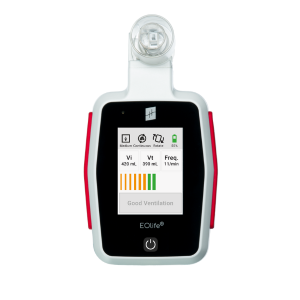
EOlife, the only Ventilation Feedback Device (VFD) to display the volume of gas reaching the patient’s lungs (Tidal Volume).
CE-marked and FDA-cleared medical device enabling real-time measurement of insufflated volume, tidal volume, and ventilation frequency. It provides visual feedback to deliver ventilation following ERC or AHA recommendations.
A solution: Ventilation Feedback Devices (VFDs)
One of the most effective ways to improve manual ventilation is by making it visible and measurable. That is exactly what devices like EOlife are designed to do. EOlife is a ventilation feedback device (VFD) that gives providers real-time data on each breath they deliver: tidal volume, ventilation rate, and leakage—all in an intuitive display suitable for use in high-pressure settings.
With EOlife, rescuers can instantly see whether they’re ventilating too much, too little, or just right. Clinical studies have shown that using a VFD significantly reduces both hyperventilation and hypoventilation, helping teams stay within the recommended guidelines.
But EOlife is not just a tool for improving clinical care. In research, it can be a powerful way to standardize ventilation quality across both study arms in a randomized trial. That means more accurate comparisons, fewer confounding variables, and stronger evidence. EOlife also records and exports ventilation data, allowing for deeper insights into how ventilation impacts outcomes—something that has been missing from most trials so far.
By making ventilation measurable, EOlife gives both clinicians and researchers a way to bring one of the most overlooked aspects of CPR under control.
Conclusion
Despite its importance, ventilation remains one of the least monitored variables in CPR research. Without reliable measurement or standardization, we risk drawing incomplete or even misleading conclusions from otherwise well-designed trials. Worse, we may miss opportunities to improve survival and neurological recovery in patients who could benefit.
The integration of ventilation feedback devices like EOlife into both clinical practice and research is not just feasible—it’s necessary. By finally giving ventilation the attention it deserves, we can raise the standard of care and generate stronger, more meaningful evidence to guide the next generation of CPR.
Are you interested in EOlife to improve your CPR research?
Learn more


